
In the vast realm of physics and chemistry, the states of matter play a crucial role in understanding the behavior of substances. Among these states, liquids and gases stand out as dynamic and ever-changing phases, each with its unique properties. This article explores the intriguing similarities between liquids and gases, shedding light on the fundamental principles that govern these two fluid states.

Similarities in Molecular Motion:
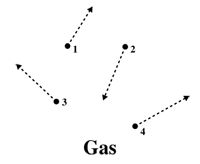
At the heart of the similarities between liquids and gases lies the concept of molecular motion. In both states, molecules are in constant motion, exhibiting translational, rotational, and vibrational movements. In liquids, molecules are closely packed, allowing for relative movement but maintaining a degree of cohesion. Similarly, in gases, molecules are more spaced out, enabling them to move freely and rapidly in all directions. This shared characteristic highlights the kinetic nature of both liquid and gas states.
Adaptability to Container Shape:
One notable similarity between liquids and gases is their ability to adapt to the shape of the container they occupy. In a liquid state, molecules have enough freedom to flow and take the shape of the container while maintaining a constant volume. Gases, too, exhibit this property, as they expand to fill the available space, conforming to the shape of the container they are in. This adaptability is a key feature that distinguishes both states from solids.
Pressure and Volume Relationship:
The relationship between pressure and volume is another shared characteristic of liquids and gases, as described by Boyle’s Law. In both states, an increase in pressure tends to decrease volume, and vice versa. This principle holds true for liquids, where compressibility is limited, as well as for gases, which can be significantly compressed or expanded based on external pressure changes. Understanding this relationship is crucial in various scientific and engineering applications.
Temperature-Dependent Changes:
Both liquids and gases undergo temperature-dependent changes in their physical properties. As temperature increases, molecules in liquids gain energy, leading to increased motion and, in many cases, a transition to the gaseous state. Similarly, gases experience changes in volume and pressure with temperature variations, as described by Charles’s Law. This temperature dependence underscores the dynamic nature of both states and their responsiveness to external energy input.
Phase Transitions:
The concept of phase transitions is applicable to both liquids and gases. While liquids can transition to gases through processes like evaporation or boiling, gases can condense into liquids through cooling or compression. This ability to transition between states emphasizes the dynamic equilibrium that exists between liquids and gases, influenced by factors such as temperature and pressure.
Real-World Applications:
The similarities between liquids and gases find practical applications in various industries and everyday life. Understanding their shared properties is crucial in fields such as thermodynamics, fluid mechanics, and chemical engineering. For example, the principles of liquids and gases are integral to the design of heat exchangers, refrigeration systems, and propulsion technologies.
In conclusion, the similarities between liquid and gas states extend beyond their fluidity and touch upon fundamental principles of molecular motion, adaptability to container shape, pressure-volume relationships, temperature-dependent changes, and phase transitions. Acknowledging these commonalities not only deepens our understanding of the physical behavior of matter but also enhances our ability to apply these principles in scientific research and technological advancements.
comparison table highlighting key similarities between liquid and gas states:
| Aspect | Liquid | Gas |
| Molecular Motion | Constant motion with translational, rotational, and vibrational movements. Molecules are closely packed. | Constant motion with translational, rotational, and vibrational movements. Molecules are more spaced out. |
| Adaptability to Container Shape | Adapts to the shape of the container while maintaining a constant volume. | Expands to fill the available space, conforming to the shape of the container. |
| Pressure and Volume Relationship | Follows Boyle’s Law – an increase in pressure decreases volume, and vice versa. | Follows Boyle’s Law – an increase in pressure decreases volume, and vice versa. |
| Temperature-Dependent Changes | Undergoes temperature-dependent changes, gaining energy with increased motion. Can transition to the gaseous state. | Undergoes temperature-dependent changes, experiencing changes in volume and pressure. |
| Phase Transitions | Can transition to a gas through processes like evaporation or boiling. | Can condense into a liquid through cooling or compression. |
| Real-World Applications | Used in various industries and everyday life, including heat exchangers and refrigeration systems. | Integral to the design of propulsion technologies and gas-based systems. |
This table provides a quick overview of the shared characteristics between liquid and gas states, emphasizing their dynamic nature and the fundamental principles that govern their behavior.
FAQ: Similarities between Liquid and Gas States
What characterizes the molecular motion in both liquids and gases?
Constant translational, rotational, and vibrational movements.
Molecules in liquids are closely packed, while in gases, they are more spaced out.
How do liquids and gases adapt to the shape of their containers?
Liquids flow to take the shape of the container while maintaining a constant volume.
Gases expand to fill the available space, conforming to the shape of the container.
What is the relationship between pressure and volume in liquids and gases?
Increase in pressure tends to decrease volume, and vice versa, following Boyle’s Law.
How do temperature-dependent changes manifest in liquids and gases?
Both undergo changes with increased temperature; molecules gain energy and can transition between states.
What are the commonalities in phase transitions between liquids and gases?
Liquids can transition to gases through processes like evaporation or boiling.
Gases can condense into liquids through cooling or compression.
What practical applications arise from the similarities between liquids and gases?
Understanding these states is crucial in thermodynamics, fluid mechanics, and applications such as heat exchangers and propulsion technologies.










Leave a Reply