The isomers are molecules with equal molecular formula, but with a different structure (structural isomerism) or different spatial orientation (stereoisomerism). The isomers have an equal number of atoms of each chemical element, but different arrangements of these atoms, and therefore – different properties.
The structural isomers have an equal molecular formula, but the connections or their order is different. The stereoisomers are molecules with the same sequence of bonded atoms and molecular formula, but with different 3D configurations/orientations of their atoms.
Anomers and epimers are two types of stereoisomers.

What is Anomer?
The anomer is a type of stereoisomer, differing in the configuration at the acetal/hemiacetal carbon atom (called also anomeric carbon). This is the carbon atom, derived from aldehyde or ketone functional group of an open-chain form of a carbohydrate molecule. Anomers are always cyclic molecules.
Anomers differ in their:
- physical properties,
- specific rotations,
- melting points.
The two anomers are named alpha (α) and beta (β), according to the configuration of the anomeric center and the reference atom.
The transformation of one anomer to the other is called anomerization. The process is reversible, however, the anomers are stable molecules. The anomerization leads to obtaining a mixture from both anomers, in which eventually is reached equilibrium. The percentage of the two anomers is specific for each sugar.
Examples for anomers are α-D-Fructofuranose and β-D-fructofuranose.
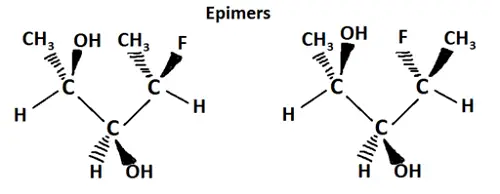
What is Epimer?
The epimer is a type of stereoisomer, differing in the configuration of any single stereogenic center. The difference is in the configuration of only one center and all the other stereocenters are with an equal configuration in each of the epimers. Epimers can be cyclic or acyclic molecules.
The chemical process of transformation of one epimer to the other is called epimerization. It can be:
- spontaneous epimerization – a relatively slow process,
- catalyzed epimerization – engendered by enzymes.
Epimers differ in their properties. Examples for epimers are the sugars galactose and glucose.
Similarities Between Anomers and Epimers
- The Anomers and Epimers are both structural isomers.
- The Anomers and Epimers both differ in one carbon atom.
- The Anomers and Epimers are both found in sugar molecules.
- The Anomers and Epimers are both formed due to the differences in the position of only one hydroxyl group.
Difference Between Anomers and Epimers
1. Definition
Anomers: The anomer is a type of stereoisomer, differing in the configuration at the acetal/hemiacetal carbon (called also anomeric carbon).
Epimers: The epimer is a type of stereoisomer, differing in the configuration of any single stereogenic center. The difference is in the configuration of only one center and all the other stereocenters are equal in each of the epimers.
2. Examples
Anomers: Examples for anomers are α-D-Fructofuranose and β-D-fructofuranose.
Epimers: Examples for epimers are the sugars galactose and glucose.
3. Carbon atom, where stereoisomerism occurs
Anomers: The carbon atom, where stereoisomerism occurs is the acetal/hemiacetal carbon (called also anomeric carbon).
Epimers: The carbon atom, where stereoisomerism occurs can be any single stereogenic center.
4. Molecule Structure
Anomers: Anomers are always cyclic molecules.
Epimers: Epimers can be cyclic or acyclic molecules

Summary:
- The Anomers and Epimers are both structural isomers, differing in one carbon atom. They are both found in sugar molecules.
- The anomer is a type of stereoisomer, differing in the configuration at the acetal/hemiacetal carbon (called also anomeric carbon).
- The epimer is a type of stereoisomer, differing in the configuration of any single stereogenic center. The difference is in the configuration of only one center and all the other stereocenters are equal in each of the epimers.
- Examples for anomers are α – D – Fructofuranose and β – D – fructofuranose; examples for epimers are the sugars galactose and glucose.
- The carbon atom, where stereoisomerism occurs in the anomers is the acetal/hemiacetal carbon (called also anomeric carbon). The carbon atom, where stereoisomerism occurs in the epimers can be any single stereogenic center.
- Anomers are always cyclic molecules, while the epimers can be cyclic or acyclic molecules.
Author: Dr. Mariam Bozhilova
Dr. Mariam Bozhilova has a Master’s degree in Ecology and PhD in Botany. Her main professional interests are in the fields of ecology, biology and chemistry. She has more than 10 years of professional experience in scientific research and environmental consultancy.







Leave a Reply