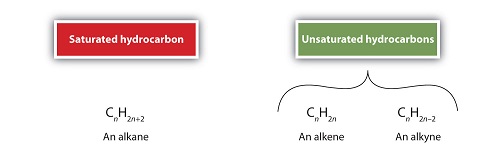
Hydrocarbons are organic compounds that are characterized by carbon and hydrogen atoms, and are often regarded as the building blocks of many organic compounds. The basic structure of these compounds is that of hydrogen atoms attached to carbon atoms. Some structures can have single or double or triple bonds. It is this variation of bonds that classifies the hydrocarbons into saturated and unsaturated hydrocarbons. The saturated hydrocarbons are composed of single bonds, whereas the unsaturated hydrocarbons are characterized by triple and double bonds. The physical properties are similar but chemical properties are different because of the variation of covalent bonds.
Definition of Saturated Hydrocarbons
As highlighted, saturated hydrocarbons are noticed by their single bonds between carbon atoms, and hydrogen atoms occupying the remaining covalent bonds on the carbon atoms. That is the main difference between the saturated and the unsaturated hydrocarbons. The carbon atom can only attach to four atoms, that is, to other carbon atoms or the hydrogen atoms. The maximum bonds it can take are 4.
The saturated hydrocarbons are the simplest organic compounds and are the least polar organic compounds. The typical saturated hydrocarbons are the alkanes, with the simplest structures known as ethane, propane, methane, butane, pentane, hexane, etc. The general formula of alkanes is CnH2n+2 where there are many hydrogen atoms than there are carbon atoms. The other typical saturated hydrocarbons include the cycloalkanes.
In combustion, the saturated hydrocarbons show a blue and non-sooty flame. They are predominantly used as fuels of automotive mobiles because of their flammability. The common fuel that is used as a cooking gas is the butane. During combustion, the saturated hydrocarbons usually yield water vapor, heat, CO2, and light. With regard to chemical properties, saturated hydrocarbons are less reactive because of their carbon-carbon single bonds. They are often exacted from animal feces and fossil plants.
Definition of Unsaturated Hydrocarbons
Unlike the saturated hydrocarbons, these organic compounds usually contain double or triple carbon-carbon covalent bonds. In double bonds, at most two hydrogen atoms can be bonded to the carbon atom while the other two bonds bond to the other carbon. And in triple bonds, only one hydrogen atom is bonded to the carbon atom while the other three bonds are covalently bonded to the other carbon atoms. Generally, there are less hydrogen atoms than carbon atoms in unsaturated hydrocarbons.
Because of the multiple bonds in unsaturated hydrocarbons, they tend to be more reactive than the single-bonded saturated hydrocarbons. In combustion, unsaturated hydrocarbons deliver a yellow and sooty flame. The types of unsaturated hydrocarbons include the alkynes, aromatic hydrocarbons and the alkenes. The common alkenes molecules include ethane, propene, pentene, etc. and alkynes include propyne, ethylene and acetylene. The aromatic hydrocarbons include benzene. The general formula for alkenes is CnH2n and for alkynes is CnH2n-2.
Key Differences between Saturated and Unsaturated Hydrocarbons
Structure
Saturated Hydrocarbons – they are characterized by a single covalent bond between carbon atoms. The carbon atom is usually bonded to four hydrogen atoms. Examples goes to CH4 (methane), C2H8 (ethane). They are all of the structure CnH2n+2.
Unsaturated Hydrocarbons – they are characterized by double or triple bonds between carbon atoms. The examples of these compounds include the alkynes, alkenes and the aromatic hydrocarbons. Alkynes include the C2H2 (ethyne), and alkenes include the C2H4 (ethane).
Reactivity
Saturated Hydrocarbons – they are less reactive because of their single bonds
Unsaturated Hydrocarbons – they are more reactive because of their multiple bonds
Combustion
Saturated Hydrocarbons – produce a blue and non-sooty flame
Unsaturated Hydrocarbons – produce a yellow and sooty flame
Origin
Saturated hydrocarbons – from fossil plants and animal matters
Unsaturated Hydrocarbons – from plants
Proportion of Hydrogen and Carbon Atoms
Saturated Hydrocarbons – have more hydrogen atoms than carbons than its unsaturated counterparts
Unsaturated Hydrocarbons – contain less hydrogen atoms than its saturated counterparts

Summary
- Hydrocarbons are organic compounds that are categorized into saturated and unsaturated compounds
- Saturated hydrocarbons have single carbon-carbon covalent bonds
- They have more hydrogen atoms than carbon atoms
- They are less reactive because of the single bonds
- Common saturated hydrocarbons are alkanes with the simplest form as methane, ethane, butane, propane
- Burn into a blue and non-sooty flame
- Unsaturated hydrocarbons have double or triple covalent bonds
- Common unsaturated hydrocarbons are alkenes, alkynes and aromatic hydrocarbons
- They are more reactive because of their multiple bonds
- They burn into a yellow and sooty flame
Author: Lusi Madisha
Lusi is a professional writer and a researcher for more than seven years. She is a chemical engineering graduate from the University of South Africa.










Leave a Reply